
Rwanda Food and Drugs Authority (FDA) has recalled sale of locally made hand sanitizers products (BEU) after producers failed to meet the markets standards.
The cancellation of the locally made product comes at a time when demand for hand sanitizers in Rwanda has increased as a result of hygiene measures to fight coronavirus infections.
In a statement released this Wednesday April 1, 2020, the authority said that the sanitizers didn’t meet the standards to be consumed by the public.
“Reference is made to laboratory quality control test results which revealed the aforementioned hand sanitizer failed to meet quality specifications,” the FDA said in part.
“Therefore in the interests of protecting the public health Rwanda FDA hereby orders recall of hand sanitizers with following description: Batch No. 090208032022, manufactured 08/03/2020, with expiry date 07/03/2022” the FDA stated.
With this, the Acting Director General Dr. Charles Karangwa said, the manufacturing plant is temporarily suspended.
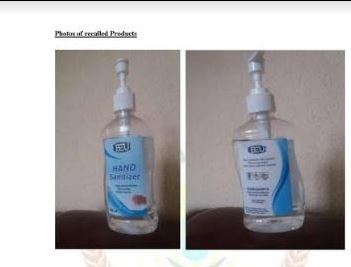 He urged users and suppliers to stop use and distribution and to return the products to the supplier for proper management.
He urged users and suppliers to stop use and distribution and to return the products to the supplier for proper management.
Karangwa also reminded the public to be cautious by checking basic information on labels to ensure quality before reception.



